In 2004, when graphene was discovered, it was hailed as a ‘wonder material’. It boasted unrivalled properties and was set to revolutionise industries. However, 18 years on, its potential is yet to be fully realised, largely due to the challenge of commercially producing the defect-free material which industry demands. Nonetheless, the eyes of academia and industry have been opened to the opportunities 2D materials afford, and unlike graphene, one material that is readily scalable, and boasts a wealth of desirable properties, is layered double hydroxide (LDH).
LDHs are layered materials with layers of positively charged mixed metal hydroxides separated by layers of negatively charged anions, creating a neutral material. The proportion and identity of the incorporated metal cations and interlayer anions are readily substituted, offering exceptional compositional flexibility and variable interlayer separation. The first LDH, hydrotalcite, was discovered in the early 19th century in a Norwegian geological specimen. Today, LDHs can be commercially synthesised as a fully synthetic material.
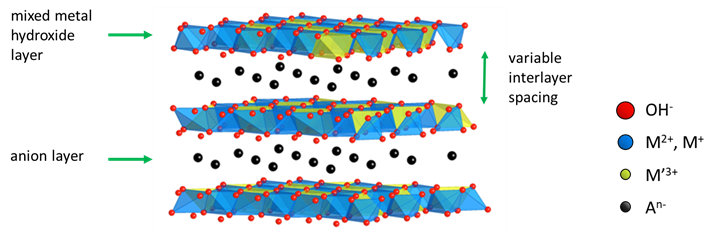
The tuneable chemistry, properties, and low cost of LDH opens the doors to a vast array of applications. For example, the high anion exchangeability provides enormous capture capacities for toxic Ag(I), Hg(II), As(II) and Cr(VI) complexes, making LDH a prime candidate for the next-generation of water purifiers.1 In medicine, the high anion exchangeability provides fantastic corrosion resistance for biomedical Mg-alloys (e.g., for stents), through Cl- scavenging.2 Moreover, the high loading efficiencies, tuneable surface charge and variable compositions allow LDHs to intercalate a range of organic molecules such as antigens, DNA, RNA, hormones and vitamins, providing a fertile ground for the development of targeted therapeutic agent delivery.3,4 LDHs are also being exploited in regenerative medicine and tissue engineering due to their thermal stability, mechanical strength, and biocompatibility.3
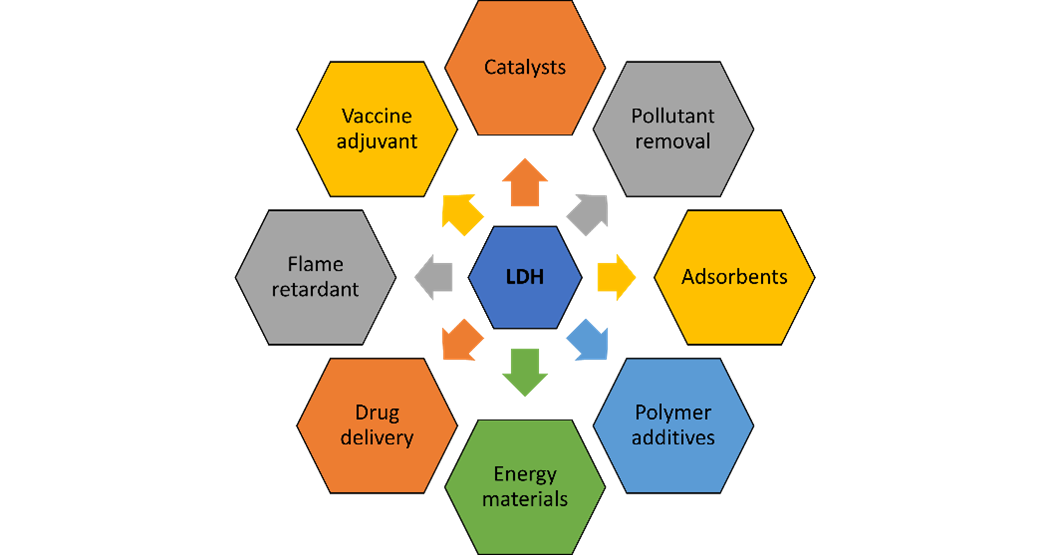
LDHs can have a wide variety of particle sizes (range of nm-µm) and morphologies (e.g., rosette, platelet), which can be tuned and controlled to meet the needs of an application. For example, high aspect ratio platelets can be selectively synthesised, which offer outstanding dual oxygen and water vapour barrier properties.5 This LDH barrier additive can be incorporated in monomaterial films, offering a recyclable alternative to multicomponent flexible packaging films. Further facilitating a greener future, LDHs can be used to enhance the thermal stability of polypropylene battery separators, delivering safer Li-ion batteries.

In summary, the high tunability, low cost, scalability, and array of desirable properties affords LDH incredible versatility, making it the ideal material for an array of applications. The number of potential applications for LDH is only growing, and in collaboration with the University of Oxford, SCG Chemicals have the unrivalled expertise and technology to unlock LDHs full potential.
LDH is a key component of the CIERRA™ platform. Want to learn more about LDH or the CIERRA™ platform? Don’t hesitate to contact our team at: info@cierra.com
References:
- Zhang L, Wang H, Zhang Q, et al. Demand-oriented construction of Mo3S13-LDH: A versatile scavenger for highly selective and efficient removal of toxic Ag(I), Hg(II), As(III), and Cr(VI) from water. Sci Total Environ. 2022;820:153334. doi:https://doi.org/10.1016/j.scitotenv.2022.153334
- Wu W, Sun X, Zhu C-L, et al. Biocorrosion resistance and biocompatibility of Mg–Al layered double hydroxide/poly-L-glutamic acid hybrid coating on magnesium alloy AZ31. Prog Org Coatings. 2020;147:105746. doi:https://doi.org/10.1016/j.porgcoat.2020.105746
- Izbudak B, Cecen B, Anaya I, Miri AK, Bal-Ozturk A, Karaoz E. Layered double hydroxide-based nanocomposite scaffolds in tissue engineering applications. RSC Adv. 2021;11(48):30237-30252. doi:10.1039/D1RA03978D
- Williams GR, Fierens K, Preston SG, et al. Immunity induced by a broad class of inorganic crystalline materials is directly controlled by their chemistry. J Exp Med. 2014;211(6):1019-1025. doi:10.1084/jem.20131768
- Yu J, Ruengkajorn K, Crivoi D-G, Chen C, Buffet J-C, O’Hare D. High gas barrier coating using non-toxic nanosheet dispersions for flexible food packaging film. Nat Commun. 2019;10(1):2398. doi:10.1038/s41467-019-10362-2

The interesting move of brand owners to Paper Packaging
By employing this new class of inorganic adjuvants, more e...

Rising demand of Lithium and its potential challenges
